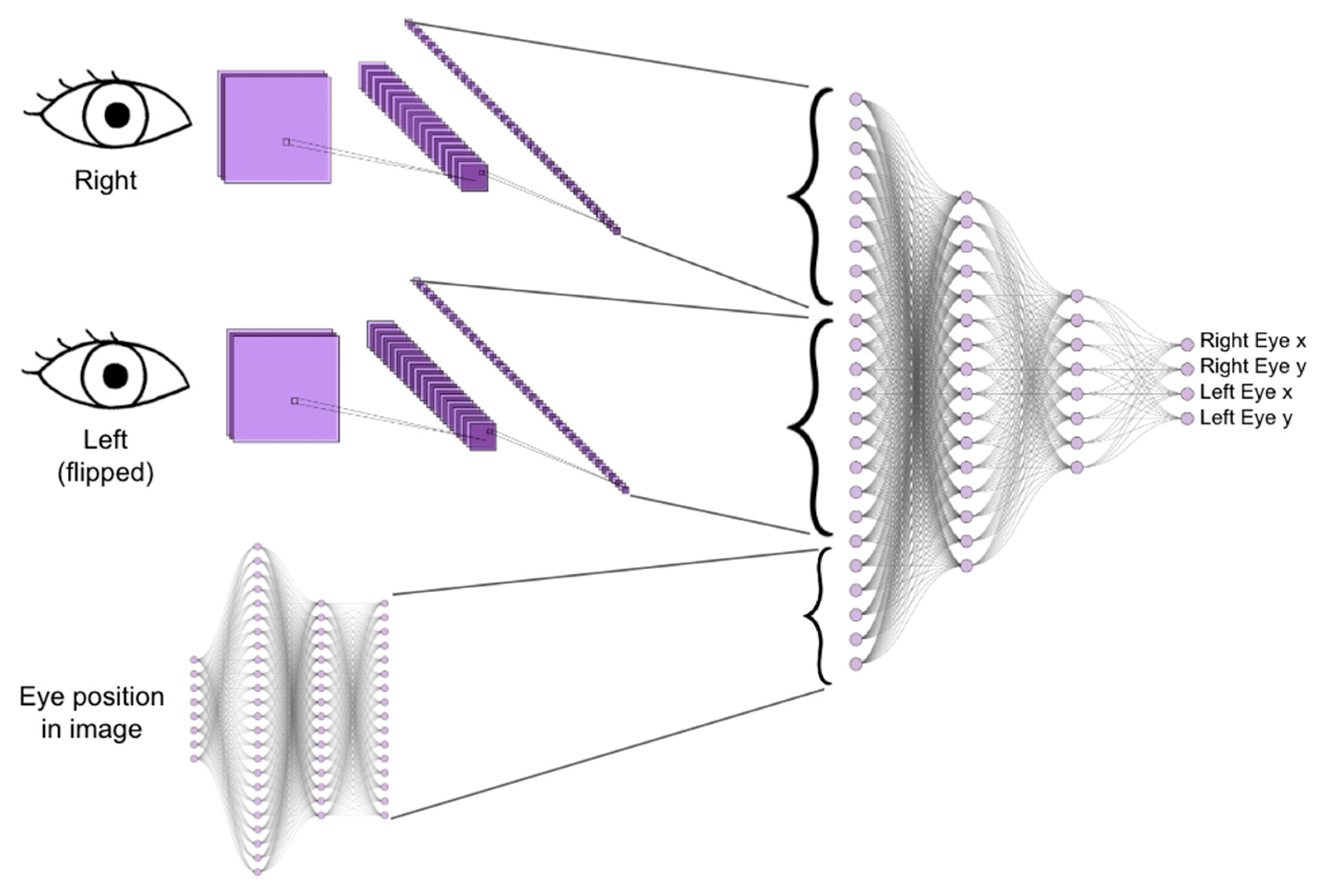
Peer-Reviewed Study Sheds Light on Viability of NeuraLight’s Oculometric Digital Biomarker Platform for Parkinson’s
As a progressive and incurable neurological condition, Parkinson’s disease has long posed challenges for patients, providers and the research community. However, recent technological advances – such as the development of NeuraLight’s oculometric digital biomarker platform – are paving the way for a paradigm shift in Parkinson’s care and clinical research. In a newly published study, NeuraLight’s technology shows promise as a tool to support diagnosis and monitor disease progression, both of which are areas of high unmet need in the management and research of neurological conditions.
Objective Assessment Unlocks New Possibilities
For decades, scientists have recognized the presence of abnormal eye movements in Parkinson’s patients. These telltale signs, which are characterized by prolonged latency of saccadic movements (Srivastava et al. 2014) and a higher error rate of anti-saccadic movements (Waldthaler et al. 2021b), have served as a window into the disease’s progression and severity. These anomalies are attributed to reduced dopamine levels, impacting the control of saccadic eye movements by the basal ganglia and their connections with the cerebral cortex, including the Superior Colliculus (SC).
The SC plays a pivotal role in both voluntary and reflexive saccades (Schiller and Stryker 1972), and its dysfunction has been linked to abnormal responses to visual stimuli in movement disorders like Parkinson’s. Parkinson’s patients often exhibit slow reaction times and reduced accuracy in their eye movements, symptoms that can be directly linked to the SC’s compromised activity (Diederich et al. 2014).
Recognizing their potential to reflect the severity of motor and cognitive symptoms (Waldthaler et al. 2019), researchers have proposed oculometric measures (OMs) as a valuable tool for clinical assessment and diagnosis of Parkinson’s patients (Termsarasab et al. 2015; Zhang et al. 2021). However, until recently, the precise correlation between OMs and established clinical assessment tools, as well as disease severity, remained unclear.
A recent study, led by Professor Ruth Djaldetti, Head of the Movement Disorders Center at Rabin Medical Center in Israel, delves into the power of oculometric measures (OMs) as a tool for assessing and monitoring Parkinson’s patients.
The Key Findings: A Game-Changer for Parkinson’s
The study examined 215 Parkinson’s patients with varying disease severity, comparing the results of NeuraLight’s technology with the conventional Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS). The results are encouraging:
- Classifying Parkinson’s Patients: Significant differences in OMs were discovered between Parkinson’s patients and healthy individuals, suggesting OMs may be a useful tool in the diagnosis or earlier detection of Parkinson’s disease.
- Monitoring Disease Progression: The study also found notable differences in OMs between patients with severe Parkinson’s and those with milder forms of the disease, suggesting that OMs can be used to monitor the advancement of Parkinson’s.
- Objective Patient Evaluation: Correlations between NeuraLight’s oculometric test results and clinical assessments were demonstrated, offering a new, objective tool for patient evaluation.
What This Means for Parkinson’s Patients
NeuraLight’s technology allows for the sensitive, stable, and objective assessment of a patient’s neurological status. This means more accurate monitoring of disease progression, leading to more tailored and effective treatments. As Professor Djaldetti highlights, “The results of this study mark an important step on our path to knowing more about our Parkinson’s patients’ disease status.”
The Future of Clinical Research
The significance of this study goes beyond patient care, intersecting with therapeutic discovery and clinical trials. NeuraLight’s technology has the potential to detect and chart patient neurodegeneration faster than current subjective clinical rating scales. This breakthrough can translate into smaller, faster, and more cost-effective clinical trials. By harnessing OMs as a novel prognostic indicator, researchers and pharmaceutical manufacturers can explore innovative screening and precision-medicine applications for Parkinson’s.
To validate the utility of this technology in Parkinson’s clinical research, NeuraLight has initiated the PALOMA (Parkinson’s Longitudinal Oculometric Measurement Assessment) trial, a first-of-its-kind multi-center longitudinal study. The first of the planned 400 Parkinson’s patients was enrolled and evaluated at the end of August, marking a significant milestone in the development of digital tools for Parkinson’s.
NeuraLight’s technology holds the promise of improving the lives of individuals battling neurological disorders and accelerating clinical research. Amidst increasing recognition of the benefits of digital tools in neurodegenerative disorders, Neuralight is taking a leading role in advancing oculometrics as a novel and innovative technology for Parkinson’s disease.
To learn more about NeuraLight’s technology, please visit NeuraLight.ai/Technology. Read the full press release here.